Newborn Hearing Screening
The scope of this page is hearing screening for children ages 0-6 months of age. Newborn hearing screening is but one part of a comprehensive Early Hearing Detection and Intervention (EHDI) program of service.
See the screening section of the Hearing Loss (Newborn) Evidence Map for pertinent scientific evidence, expert opinion, and client/caregiver perspective.
Newborn hearing screening is the standard of care in hospitals nationwide. The primary purpose of newborn hearing screening is to identify newborns who are likely to have hearing loss and who require further evaluation. A secondary objective is to identify newborns with medical conditions that can cause late-onset hearing loss and to establish a plan for continued monitoring of their hearing status (Joint Committee on Infant Hearing [JCIH], 2007). The EHDI guidelines include hearing screening completion by 1 month of age, diagnosis of any hearing loss by 3 months of age, hearing aid selection and fitting within 1 month of confirmation of hearing loss if parents choose that option, and entry into early intervention (EI) services by 6 months of age.
In 2014, 96.1% of babies born in the United States had their hearing screened before 1 month of age (Centers for Disease Control and Prevention [CDC], 2016), and 6,163 infants were diagnosed with permanent hearing loss.
Screening programs target permanent childhood hearing loss (PCHL) irrespective of type. However, some protocols are more effective at identifying types and degrees of hearing loss within different populations (i.e., well-baby nursery or neonatal intensive care unit [NICU]).
Passing a screening does not mean that a child has normal hearing across the frequency range. Because minimal and frequency-specific hearing losses are not targeted by newborn hearing screening programs, newborns with these losses may pass a hearing screening. Because these losses have the potential to interfere with the speech, language, and psychoeducational development of children (Yoshinaga-Itano, DeConde Johnson, Carpenter, & Stredler-Brown, 2008), monitoring of hearing, speech, and language milestones throughout childhood is essential.
Roles and Responsibilities
Roles and Responsibilities of Audiologists
Audiologists, by virtue of academic degree, clinical training, and license to practice, are qualified to provide guidance, development, implementation, and oversight of newborn hearing screening programs. See ASHA's Scope of Practice in Audiology (ASHA, 2018).
Appropriate roles and responsibilities for audiologists include the following:
- Providing management and oversight to other trained professionals who are performing screenings.
- Selecting screening protocols for both the NICU and the well-baby nursery.
- Selecting equipment.
- Performing hearing screenings.
- Training and monitoring (on a continual basis) screening personnel competencies.
- Selecting or developing educational materials.
- Monitoring key indicators (refer rates, miss rates, etc.).
- Developing and implementing written policies and procedures on
- infection control;
- screening techniques and process;
- documentation of screening results per medical facility protocol (e.g., medical records, electronic health record, birth certificate, discharge summary); and
- calibration and upgrade of equipment.
- Communicating screening results to families, primary care physicians, state EHDI system, and diagnostic audiology centers, as indicated.
- Providing counseling and education.
- Referring infants for audiologic and medical services, as indicated.
- Advocating for the communication needs of all individuals, including advocating for the rights and funding of services for those with hearing loss.
As indicated in the Code of Ethics (ASHA, 2023), audiologists who work in this capacity should be specifically educated and appropriately trained to do so.
Roles and Responsibilities of Speech-Language Pathologists
Speech-language pathologists (SLPs) who work in birthing hospitals may also be called upon to perform newborn hearing screenings. SLPs play a role in the hearing screening, speech/language/communication assessment, and rehabilitation of individuals with hearing loss and the referral of individuals suspected of having hearing loss to an audiologist. See ASHA's Scope of Practice in Speech-Language Pathology (ASHA, 2016).
Appropriate roles for SLPs in hearing screening programs for children ages 0-6 months include the following:
- Performing newborn hearing screenings using automated equipment in the hospital and as part of outpatient screening programs with appropriate training and oversight from the managing audiologist.
- Communicating screening results to families, including recommendations for follow-up consistent with JCIH recommendations.
- Providing counseling and education.
- Documenting and sharing screening results with state public health agencies, primary care physicians, and diagnostic audiology centers as required by the state EHDI program.
- Collaborating with audiologists, physicians, nurses, and/or other professionals to ensure timely follow-up.
- Referring infants for comprehensive audiologic, medical, and/or other professional services, as indicated.
- Advocating for the communication needs of all individuals, including advocating for the rights and funding of services for those with hearing loss.
As indicated in the Code of Ethics (ASHA, 2023), SLPs who work in this capacity should be specifically educated and appropriately trained to do so.
General Considerations
Universal newborn hearing screening (UNHS) programs typically include
- parent/guardian written education materials;
- hearing screening protocols using objective physiological test(s);
- a process for obtaining parent/guardian consent in accordance with state and federal guidelines;
- a process for communicating screening results;
- a follow-up system for infants who do not pass inpatient hearing screening or who are missed by the inpatient screening program – to include repeat screening, as indicated, or referral for appropriate evaluation and early intervention;
- documentation and data systems to track screening and follow-up;
- access to interpreters who use languages represented by the population serviced; and
- a quality assurance process to evaluate the effectiveness of newborn hearing screening (JCIH, 2007; New York State Department of Health, Early Intervention Program, 2007).
A state policy of performing screening without obtaining parent/guardian permission, or at least informing parents/guardians about screening, may violate constitutional protections of the parental/guardianship role (Berge, 1992; Clayton, 1992; Fleischman, Post, & Dubler, 1994). Although many hospitals have global consents for all newborn hearing screening procedures, screeners and personnel should be aware of hospital and state regulations regarding parent/guardian refusal.
See the ASHA State-by-State resource for more information.
Screener Roles and Characteristics
Various personnel may perform newborn hearing screenings. A screener may be an audiologist, an SLP, a nurse, a paraprofessional, or a trained volunteer. All screeners are trained in, and competent to use, the technology and protocol specific to the screening program. It is important for the screener to have a familiarity with JCIH guidelines, EHDI process, and hospital/clinic protocols, including standard precautions and patient confidentiality standards (ASHA, 2013).
Timing of Screening
Newborns cared for in the well-baby nursery are screened as close to hospital discharge as possible and prior to 1 month of age. NICU newborns are screened when they are ready for discharge and/or when they are medically stable.
Newborns who have initially passed a hearing screening are rescreened if readmitted to the hospital in the first month of life or if risk factors for hearing loss develop during the infant's hospital stay following the initial screening. State laws and hospital protocols may vary regarding which hospital is responsible for screening newborns who are transferred from one hospital to another.
Testing Environment
Screening can be done in a nursery or a quiet room with the infant resting quietly or sleeping. A sound booth is not needed. The preferred method for testing is to have the newborn resting quietly in their bassinette – although, if needed, the newborn can be held.
Pass/Refer Indications
A newborn must pass the screening in both ears during one session for the screening to be considered a "pass." Otherwise, the newborn will be referred for rescreening. If the newborn does not pass in one ear, both ears must be rescreened. If the newborn passes the screening or the rescreening and has no risk factors for late-onset or progressive hearing loss, then the screening is complete. If the newborn passes the screening or the rescreening and has risk factors for late-onset or progressive hearing loss, then it will be very important to monitor the newborn's hearing during early childhood (Harlor & Bower, 2009; JCIH, 2007).
Care should be taken not to screen newborns more times than recommended in the protocol. The probability of an erroneous "pass" outcome (i.e., of infants with hearing loss passing the screening) increases with every screen.
Standard Universal Precautions
All procedures must ensure the safety of the patient and clinician and adhere to universal health precautions (e.g., prevention of bodily injury and transmission of infectious disease). Decontamination, cleaning, disinfection, and sterilization of multiple-use equipment before reuse must be carried out according to facility-specific infection control policies and procedures and according to the manufacturer's instructions (Siegel, Rhinehart, Jackson, Chiarello, & Healthcare Infection Control Practices Advisory Committee, 2007).
Technology
Auditory brainstem response (ABR) and otoacoustic emissions (OAEs) are appropriate physiologic measures for screening the newborn population. Both are noninvasive and available in automated versions that are easily utilized by trained hospital staff.
Both ABR and OAE technologies will miss delayed-onset hearing loss, mild hearing loss, or hearing loss that is present only at isolated frequencies. Both ABR and OAE responses are affected by outer or middle ear dysfunction. When a transient middle ear condition is present, both technologies will likely result in the newborn not passing the screening. Both OAE and ABR screening reflect physiologic processes within the auditory system and identify hearing loss most accurately from 2kHz to 4kHz.
Automated technologies (those that determine pass/refer) do not require interpretation. Automated screening equipment often has test parameters set by the manufacturer, and, therefore, different equipment may yield different screening results (New York State Department of Health, Early Intervention Program, 2007).
Even if diagnostic (i.e., nonautomated) technology is used, and an audiologist is interpreting the results, procedures in the nursery are limited to screening (pass/refer). Equipment that is used should be calibrated and maintained according to the manufacturer's specifications.
The factors that influence the selection of screening technology include
- the population to be screened (i.e., well-baby nursery vs. NICU);
- who will be conducting the screening (i.e., audiologists vs. trained technicians);
- the size of the screening population;
- the time required of the screener; and
- the cost (e.g., equipment purchase and calibration, disposable equipment).
Auditory Brainstem Response (ABR)
ABR activity is a direct measurement of the neural response to sound that is generated along the auditory system from the level of the cochlea and through the VIII nerve and pontine-level of the brainstem and that correlates with behavioral hearing measures in the mid- to high-frequency region. The ABR is recorded using surface electrodes that are attached to the infant's head. Click stimuli are presented through insert or muff-style earphones that are worn on both ears. ABR measurements are sensitive to neural auditory disorders (i.e., auditory neuropathy). Auditory neuropathy is a neural hearing loss that leaves cochlear (outer hair cell) function intact. It is more prevalent in the NICU population than in the well-baby nursery population (D'Agostino & Austin, 2004). ABR screening is less sensitive to outer ear debris than OAE screening, resulting in lower referral rates.
Stimuli
Most automated equipment presents click stimuli at 35 dB nHL at a rate of 30-37 clicks per second. If the equipment allows a choice of stimulus levels, the screening program audiologist can adjust to lower click levels.
Response Criteria
For automated ABR (AABR), manufacturers use their own proprietary stopping rule, based on a template comparison or statistical algorithms. Most instrumentation does not allow operators to change the stopping rule criteria.
Procedure
The infant should be asleep or resting quietly for the test and positioned to reduce muscle artifact. The screener visually inspects the outer part of the ear canal to ensure that the canal is clear of debris and places the transducer. Both ears are screened during each session.
Otoacoustic Emissions (OAE)
OAEs – either transient-evoked OAEs (TEOAEs) or distortion product OAEs (DPOAEs) – are measured using a sensitive probe microphone inserted into the infant's ear canal. OAEs are a direct measure of outer hair cell and cochlear function in response to acoustic stimulation. They yield an indirect estimate of peripheral hearing sensitivity. OAEs are not sensitive to disorders central to the outer hair cells, such as auditory neuropathy. OAEs will be absent when there is outer or middle ear dysfunction or debris/blockage in the ear canal.
Stimuli
TEOAEs use a high-level click, approximately 80 dB pSPL, and a subtraction (sometimes referred to as nonlinear) paradigm to reduce stimulus artifact. DPOAEs use mid-level stimuli (f1 primary = 65 dB SPL and f2 primary = 55 dB SPL).
Response Criteria
Many manufacturers program response criteria into the OAE unit. At least three test frequencies – of 2000 Hz, 3000 Hz, and 4000 Hz – are evaluated during the screening. Usually, signal-to-noise ratios (SNRs) of at least 6 dB are used; however, some manufacturers will set their own SNRs. DPOAE SNRs vary, depending on whether mean SNR is calculated or mean plus 1 or 2 standard deviations of noise. In addition, a minimum absolute DPOAE level of 5 dB SPL is imposed.
- TEOAEs: Typically, a minimum of 50 averages is collected before testing is terminated.
- DPOAEs: DPOAEs are often terminated based on SNR. Rather than a minimum number of averages, manufacturers choose a minimum length of averaging time.
Procedure
The infant should be asleep or resting quietly for the test and positioned to reduce muscle artifact. A snug probe fit is essential for valid and reliable recordings. Ears should be screened one at a time, with the infant placed on their side and the ear being screened facing up. The screener visually inspects the outer part of the ear canal to ensure that the canal is patent and clear of debris. Prior to insertion of the probe, a gentle massage of the area below the tragus helps to open a collapsed canal or dislodge debris that may be blocking the canal.
In OAE screenings, the stimulus level is calibrated in each ear according to the manufacturer's specifications. After stimulus-level requirements have been met, OAEs are collected to meet stopping criteria. If OAEs do not appear to be present, the probe is taken out and inspected to determine if the probe is blocked with cerumen or vernix. A blocked probe should be cleaned and reinserted, and the screening should be repeated.
Protocols
Screening protocols can be broadly classified into four different categories:
- ABR only
- OAEs only
- Two-tier (OAEs with immediate ABR rescreening if OAE is not passed)
- Two-technology (ABR and OAEs)
The choice of protocol for a newborn hearing screening program is based on the specific needs of the population (i.e., well-baby nursery or NICU) and requirements of the state or hospital program.
Screening with ABR Only
ABR can be used in both the NICU and the well-baby nursery. The rationale for an "ABR-only" protocol is that both neural and cochlear hearing losses will be detected using one type of technology. ABR screening is recommended by the JCIH (2007) for newborns cared for in the NICU for more than 5 days because they are at greater risk for neural hearing loss.
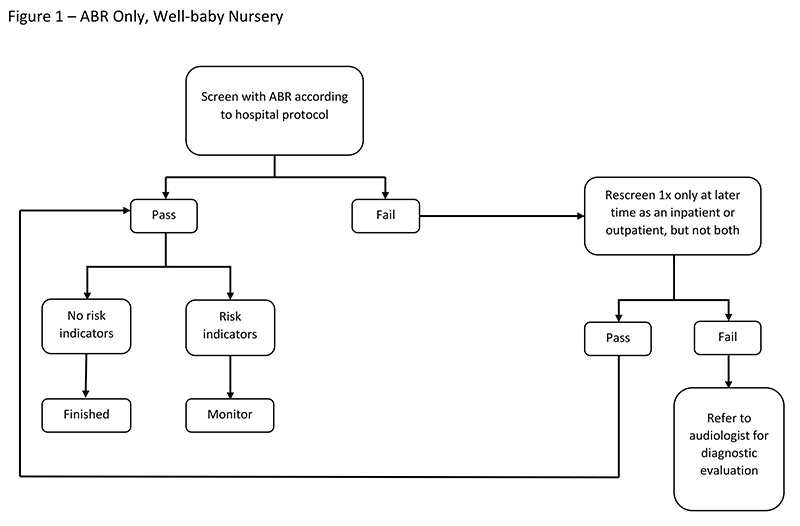
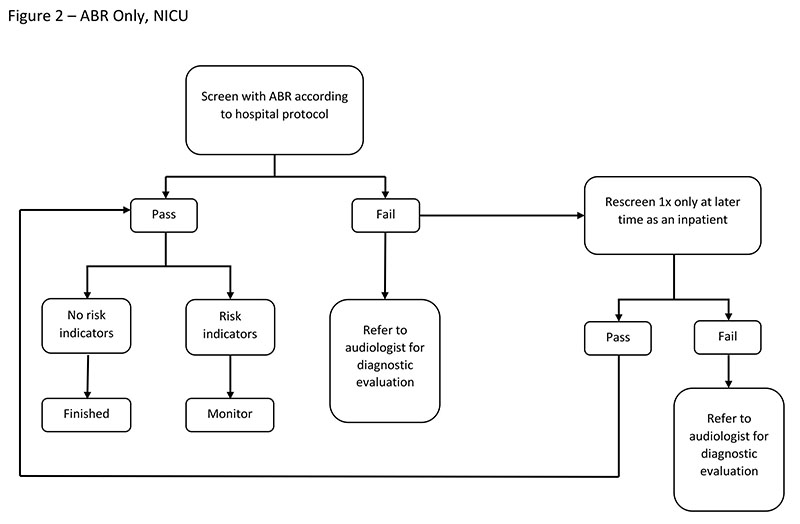
Factors To Consider
Consider the following factors when using an ABR-only screening approach:
- More of the auditory system is accessed with ABR screening than with OAEs, allowing for detection of neurologic involvement.
- ABR screening results are less susceptible to false positives due to ear canal debris than are OAE screenings (Vohr et al., 2001).
- ABR screening can be less cost effective than an OAE screening because of the higher cost of disposables (electrodes and disposable earphones) and increased personnel time (Berg, Prieve, Serpanos, & Wheaton, 2011). This cost differential may be balanced with a lower refer rate, resulting in reportedly lower or similar overall program costs (Vohr et al., 2001).
- ABR test time can be longer than OAE test time because of electrodes application (Berg et al., 2011).
- ABR will result in false positives when used with babies who have immature neurological systems that affect the ABR waveform (Turchetta et al., 2012).
Screening With OAE Only
Otoacoustic emissions – either transient-evoked OAEs (TEOAEs) or distortion product OAEs (DPOAEs) – are recommended for use in well-baby nurseries. Because OAEs are sensitive to outer ear debris and middle ear fluid that may be present at birth, most OAE screening protocols involve an outpatient rescreening of those newborns who do not pass the screening at hospital discharge.
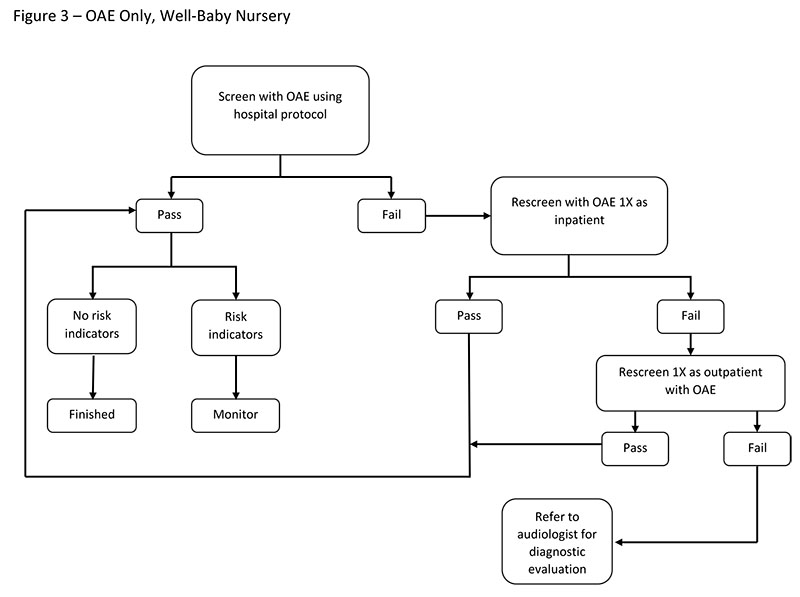
Factors To Consider
Consider the following factors when using an OAE-only screening approach:
- OAE disposable supplies are often less expensive than those used with ABR.
- OAE test time has been reported to be shorter than ABR test time (Berg et al., 2011).
- OAEs do not detect neural hearing losses.
- OAEs have a higher inpatient refer rate as compared to ABR. Outpatient rescreening is advised for newborns who do not pass inpatient screening before referral to an audiologist for diagnostic testing.
- OAEs may be reduced or absent due to outer ear debris and/or middle ear fluid common in the newborn population, resulting in higher refer rates. That is, OAEs have a higher false positive rate than ABR.
Two-Tier Screening: OAE Followed by ABR if the OAE is Not Passed
To provide options for newborn hearing screening and to reduce overall program costs, a two-tier screening approach can be used. In this approach, OAE screening is completed on both ears first, followed in the same session by an ABR only for those newborns who do not pass the OAE screening. Each ear must pass the OAE screening to be considered a "pass." If the OAE screening is not passed in one or both ears, an ABR screening test is performed. If the infant passes the ABR, the baby has "passed" the hearing screening. If one or both ears do not pass the ABR, the infant is referred for outpatient diagnostic testing.
The rationale behind a two-tier approach is that OAEs have lower disposables cost and are faster to perform than ABR. However, OAEs have a higher refer rate than ABR. Conducting an OAE screening first reduces the number of newborns who require an ABR screening.
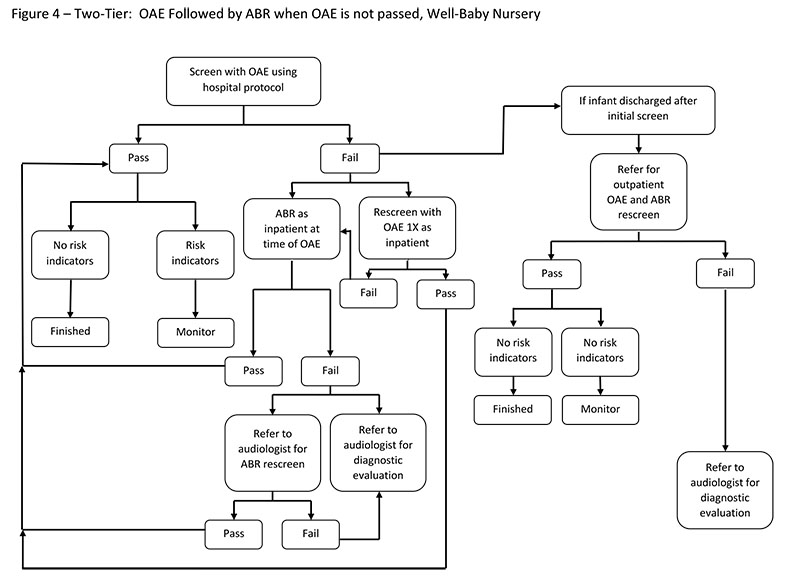
Factors To Consider
Consider the following factors when using a two-tier screening approach:
- The number of referrals for diagnostic testing will be lower in a two-tier protocol than in an OAE-only protocol.
- Two types of disposables will be required, and two pieces of equipment may be needed, although some manufacturers have both tests on one piece of equipment.
- Personnel must be trained on two procedures.
- Because tests differ in detecting different degrees of hearing loss, a baby may not pass the OAE screening but pass the ABR screening (Johnson et al., 2005). As a result, some children with mild hearing loss may be missed.
- A two-tier protocol may miss auditory neuropathy because infants who pass an OAE screening will not be referred for an ABR.
Two-Technology Screening: ABR and OAE
In a two-technology protocol, newborns must pass both an OAE screening and an ABR screening. Both ears are tested with ABR and OAEs, and each ear must pass both tests to be considered a "pass." Although pass/refer rates are available for this protocol in the NICU (Berg, Spitzer, Towers, Bartosiewicz, & Diamond, 2005) and in the well-baby nursery (Berg et al., 2011), no sensitivity or specificity data are available.
If a newborn does not pass the screening for one or both technologies, they can be rescreened with that technology (or technologies) one time only: either before hospital discharge or in an outpatient setting. If the newborn does not pass one or both of the second screenings in one or both ears, the newborn is referred for outpatient diagnostic testing.
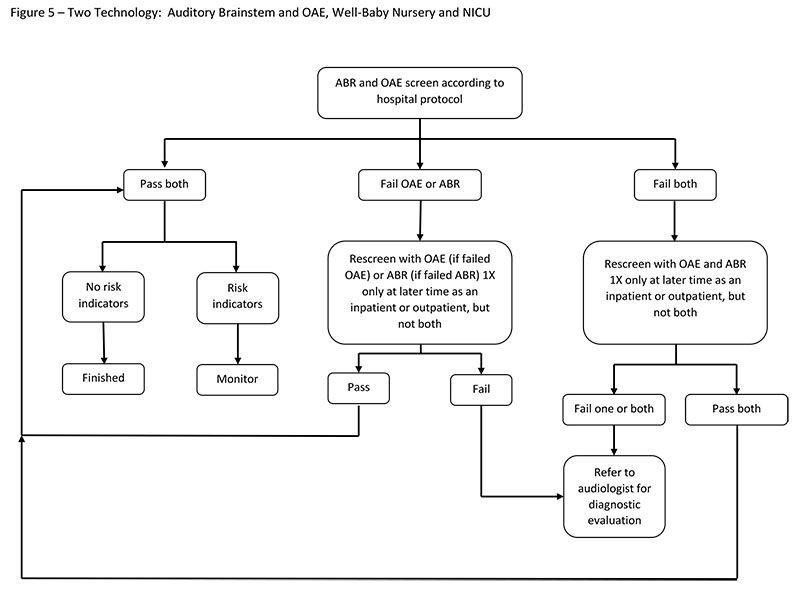
Factors To Consider
Consider the following factors when using a two-technology screening approach:
- A two-technology protocol is more likely to detect both mild hearing loss (due to the use of OAE technology) and neural hearing loss (due to the use of ABR technology).
- Two pieces of equipment or a device that includes both tests must be purchased and maintained, as well as two types of disposables.
- Personnel must be trained on both technologies.
- Additional screening time is required (Berg et al., 2011) because two procedures are completed.
- Refer rates may be higher because two screening tests must be passed (Berg et al., 2005; Kirkim, Serbetcioglu, Erdag, & Ceryan, 2008).
Detection of Late-Onset Hearing Loss
The American Academy of Pediatrics (AAP) recommends that every newborn have a medical home to provide coordinated, comprehensive, primary care that is accessible and family centered. The medical home plays a key role in supporting EHDI systems and detecting late-onset hearing loss (Mehl & Esquivel, 2016). Although initial newborn hearing screenings are not typically conducted in the medical home, the JCIH recommends that the pediatric primary care provider regularly monitor all infants and children for hearing loss and communication development in the medical home (JCIH, 2007). This allows for the identification of children with either missed newborn, late-onset, or progressive hearing loss, regardless of the presence or absence of high-risk indicators at birth. The onset of hearing loss can occur at any time in a child's life. Developmental milestones, hearing skills, and speech and language milestones should be monitored in all children, consistent with the Bright Futures/AAP periodicity schedule [PDF]. See the Childhood Hearing Screening Practice Portal page for more information on this topic.
Documentation and Dissemination
Newborn hearing screening documentation requirements are based on hospital and state mandates or protocols and can include the recording of screening results into the medical record, electronic health record, birth certificate, discharge summary, or state EHDI data system. In addition, screening results must be provided to the family and the newborn's physician. Patient records must be in full compliance with the Health Insurance Portability and Accountability Act (HIPAA, 1996).
Documentation of newborn hearing screening typically includes
- child's name and date of birth;
- parent/guardian contact information;
- date and time of all screenings;
- type of screening (OAE, ABR, or both);
- outcome of the screening for each ear;
- recommendations for next steps;
- follow-up information, if applicable (e.g., appointment date/time/place, phone number of the follow-up facility);
- hospital transfer information, if applicable; and
- signature of personnel conducting the screening.
Collecting parent/guardian and pediatrician contact information is critical in the effort to prevent loss to follow-up, especially for those newborns who do not pass the screening. To track newborn screenings effectively, states stipulate which information will be reported to the appropriate government agency (e.g., state EHDI program). See the ASHA State-by-State resource for a summary of requirements.
Counseling and Education
Providing appropriate counseling and education is an important part of the newborn hearing screening process.
Parent/Guardian Education
Parent/guardian education regarding newborn hearing screening begins before an infant is screened. If information about the screening process is provided only after a mother is admitted to the hospital to deliver their child, they may not have the level of concentration needed to consider the information being presented. Educational materials can be included in patient education packets, as part of hospital prenatal education programs, and in public health clinic outreach programs. Materials should be written in plain language, culturally appropriate, and available in the preferred language of the family. Information should also be conveyed orally in the preferred language of the parents/guardians.
Educational materials typically include information on
- the importance of early hearing detection and intervention;
- an overview of the screening process, including assurances that screening is safe and noninvasive;
- the meaning of a screening "pass" and "refer";
- the importance of follow-up after screening;
- the steps involved in declining screening, if desired;
- risk factors for late-onset hearing loss; and
- developmental milestones for typically developing children.
Communicating Screening Results
Where possible, audiologists are the professionals who communicate with the family when an infant has not passed a hearing screening. When an audiologist is not available to convey results, other qualified individuals (e.g., SLPs, nurses, technicians, physicians) must be available to provide and explain screening outcomes.
When communicating screening results, consider that
- communication is confidential and should be presented in a family-focused manner;
- information should be delivered in a clear and concise manner that avoids technical jargon;
- information should be provided in the family's preferred mode of communication and language;
- findings should be presented in a positive manner, emphasizing the importance of appropriate follow-up; and
- parents/guardians should be given the opportunity and encouraged to ask questions.
If an infant is referred for further testing, the audiologist provides the information in such a way that the family feels supported yet clearly understands the importance of following up with further audiologic testing. If follow-up is needed, families should receive
- explicit recommendations on how to secure follow-up testing;
- contact information for an audiologist whom they can contact directly with questions in the interim; and
- available local, state, and national resources that they can use to obtain information about subsequent stages of the EHDI process.
Families of newborns who pass the hearing screening should be counseled about the need for ongoing surveillance and periodic monitoring.
Loss to Follow-up/Loss to Documentation
In general, a baby who does not complete the recommended diagnostic or intervention process is designated as "lost to follow-up" (LTF). " Lost to documentation" (LTD) designates those infants who did not pass their hearing screening and whose diagnostic or intervention status has not been reported to the EHDI program.
There has been a slow but steady decline in the U.S. LTF/LTD rates. In 2014, 34.4% of the infants who did not pass their final newborn hearing screening did not complete follow-up and were categorized as LTF/LTD (CDC, 2016).
Populations at particular risk for LTF include infants born in home births, border babies (babies who live in one state but are born in another), and babies born in one hospital and transferred to another. The medical home plays a major role in the care of infants who do not pass or do not receive a newborn hearing screening by helping families understand the EHDI process and encouraging prompt follow-up. The medical home can ensure that appropriate and timely steps are taken to identify children who are deaf/hard of hearing through rescreening and referrals for diagnostic evaluations and early intervention services (CDC, 2015; Mehl & Esquivel, 2016).
The National Initiative for Children's Healthcare Quality (NICHQ) identified several strategies to decrease loss to follow-up for infants who do not pass their hearing screening, including the following:
- Scripting the communication with parents/guardians regarding the screening result.
- Standardizing the process for collecting contact information at the time of the screening, including obtaining a second point of contact for the family and verifying the primary care physician (PCP) or clinic responsible for follow-up.
- Scheduling a follow-up appointment (rescreening or diagnostic) before the family leaves the hospital and stressing its importance to the family.
- Calling the family to verify the follow-up appointment and provide assistance, such as transportation vouchers if necessary.
- Using fax-back forms between all parts of the care compendium (audiology, PCPs, etc.) to ensure that the results and need for follow-up are understood by all parties.
- Obtaining consent from parents/guardians for release of information at first contact with early intervention, so that information can be shared between early intervention, the PCP, and the state EHDI database (Russ, Hanna, DesGeorges, & Forsman, 2010).
System Issues
There are ongoing efforts to improve EHDI systems so that babies who do not pass their newborn hearing screening are not LTF or LTD.
These efforts may include the following:
- Requiring hospital personnel to identify the newborn's medical home/primary care physician or clinic (Health Resources and Services Administration [HRSA], 2009)
- Using designated physicians as AAP EHDI Chapter Champions to educate medical home providers and respond to issues regarding newborn hearing screening in the state (AAP, 2017)
- Increasing parent/guardian education prior to screening, including the details of the hearing screening and follow-up and the urgency of early diagnosis of hearing loss (Alexander & Van Dyck, 2006)
- Improving coordination, integrated data management, and tracking systems among service providers
- Encouraging audiologists to report diagnostic results as recommended and as required by state regulations
- Obtaining required consent for release of information at first contact with early intervention so that information can be entered into an EHDI database
- Maintaining consistent, stable state and federal funding for EHDI programs
- Increasing the number of hospitals that involve audiologists in the newborn hearing screening program
Program Evaluation and Quality Assurance
Various quality indicators and benchmarks can be used to evaluate quality assurance and performance relative to screening and diagnosis, including the following (JCIH, 2007):
- The number of newborns who complete screening (inpatient and outpatient) by 1 month of age. The benchmark is 95%.
- The number of newborns referred for diagnostic audiologic evaluation. The benchmark is 4%.
- The number of newborns who did not pass the screening and went on to have a comprehensive diagnostic audiologic evaluation by 3 months of age. The benchmark is 90%.
- The percentage of infants obtaining amplification within 1 month of hearing loss confirmation for families choosing that option. The benchmark is 95%.
Other quality assurance indicators may include
- the number of follow-up appointments scheduled and recorded;
- parent/guardian satisfaction with the process;
- timeliness and accuracy of screening results; and
- the capacity to analyze and report data.
States can also evaluate their programs as they report the statewide findings to the CDC. CDC data collection includes the number of
- live births;
- newborns screened;
- missed screenings (including home births and transferred newborns);
- newborns not screened due to parent/guardian refusal;
- newborns passing the screening prior to discharge;
- newborns discharged not passing screening in one or both ears;
- infants referred for diagnostic testing;
- infants who had inconclusive results;
- infants transferred in/out of the facility;
- infants screened who were transferred in/out of the facility;
- deceased infants;
- newborns passing outpatient (re)screening; and
- newborns not passing outpatient screening in one or both ears.
Laws and Regulations
Privacy regulations – including HIPAA, Family Education Rights and Privacy Act (FERPA), Part C Privacy Regulations (Individuals with Disabilities Education Act [IDEA]), and state privacy reporting laws – may affect the sharing of information among service providers. Audiologists and other EHDI stakeholders must understand and abide by these regulations and develop procedures to ensure that information is shared in a timely way to avoid LTF. See the National Center for Hearing Assessment and Management (NCHAM, 2008) white paper, Impact of Privacy Regulations [PDF]. Regardless of whether or not a consent form is required, a parent/guardian signature can be used to document that the family has been informed of the screening results and next steps. Audiologists are responsible for understanding how privacy laws are interpreted in their specific states or programs.
All 50 states and the District of Columbia have EHDI programs established either by law or by voluntary compliance. See the ASHA State-by-State resource for more information.
Reimbursement and Program Funding
Newborn hearing screening funding varies from state to state. States procure funds from fees, Medicaid, the Title V Maternal and Child Health Block Grant program, and state general revenues. The trend is to identify funding sources other than grants to ensure program continuity should grant support become unavailable. Costs associated with newborn hearing screening include those associated with the tests (equipment, disposables, staff time) and those associated with program management (data entry, data analysis, follow-up activities, outcomes monitoring).
Early Periodic Screening, Diagnostic, and Treatment (EPSDT) Program
Through the EPSDT program, a set of services and benefits are mandated for all individuals under age 21 years who are enrolled in Medicaid. Federal rules encourage partnerships between state Medicaid and Title V agencies to ensure better access to and receipt of the full range of screening, diagnostic, and treatment services. EPSDT funds are an appropriate reimbursement source for screening and diagnostic services.
Current Procedural Terminology (CPT) Codes
There are a number of CPT codes used to describe early hearing detection testing to a payer. Codes are defined by the payer or program. For specific instructions, see NCHAM State Contacts.
ASHA Resources
- ASHA State-by-State
- Audiology Information Series: Newborn Hearing Screening [PDF]
- Early Hearing Detection and Intervention (EHDI)
- EHDI Phase II State Model Legislation [PDF]
- Hearing Screening: Newborns and Infants
- Model Universal Newborn/Infant Hearing Screening, Tracking, and Intervention Bill
- Flow Charts
Other Resources
This list of resources is not exhaustive and the inclusion of any specific resource does not imply endorsement from ASHA.
- American Academy of Pediatrics: Early Hearing Detection and Intervention (EHDI)
- American Academy of Pediatrics: EHDI Guidelines for Pediatric Medical Home Providers [PDF]
- American Academy of Pediatrics: EHDI Program Chapter Champion Job Description [PDF]
- American Academy of Pediatrics: Medical Home Rescreening Guidelines [PDF]
- Boys Town National Research Hospital: My Baby's Hearing
- Bright Futures/American Academy of Pediatrics Periodicity Schedule
- CDC: Doctors and Infant Hearing
- CDC: Hearing Loss in Children
- Health Resources and Services Administration: Enhancing Communication: Improving Care for Infants With Hearing Loss [PDF]
- Health Resources and Services Administration: Title V Maternal and Child Health Services Block Grant Program
- Joint Committee on Infant Hearing
- National Center for Hearing Assessment and Management (NCHAM)
- NCHAM: The Impact of Privacy Regulations [PDF]
- NCHAM: Medical Home
- NCHAM: Interactive Web-Based Newborn Hearing Screening Training Curriculum
- NCHAM: Resources
- NCHAM State Contacts
- U.S. Preventative Services Task Force: Hearing Loss in Newborns: Screening
Alexander, D., & Van Dyck, P. C. (2006). A vision of the future of newborn screening. Pediatrics, 117(Suppl. 3), S350-S354.
American Academy of Pediatrics. (2017). Early Hearing Detection and Intervention (EHDI). Retrieved from https://www.aap.org/en-us/advocacy-and-policy/aap-health-initiatives/PEHDIC/pages/early-hearing-detection-and-intervention.aspx
American Speech-Language-Hearing Association. (2016). Scope of practice in speech-language pathology [Scope of practice]. Available from www.asha.org/policy/.
American Speech-Language-Hearing Association. (2018).Scope of practice in audiology [Scope of practice]. Available from www.asha.org/policy/.
American Speech-Language-Hearing Association. (2023).Code of ethics [Ethics]. Available from www.asha.org/policy/.
Berg, A. L., Prieve, B. A., Serpanos, Y. C., & Wheaton, M. A. (2011). Hearing screening in a well-infant nursery: Profile of automated ABR-fail/OAE-pass. Pediatrics, 27, 269-275.
Berg, A. L, Spitzer, J. B., Towers, H. M., Bartosiewicz, C., & Diamond, B. E. (2005). Newborn hearing screening in the NICU: Profile of failed auditory brainstem response/passed otoacoustic emission. Pediatrics, 116, 933-938.
Berge, P.H. (1992). Setting limits on involuntary HIV antibody testing under Rule 35 and state independent medical examination statuses. Florida Law Review, 44, 767-805.
Centers for Disease Control and Prevention. (2015). Doctors and infant hearing. Retrieved from https://www.cdc.gov/features/infant-hearing/
Centers for Disease Control and Prevention. (2016). Summary of 2014 national EHDI data. Retrieved from https://www.cdc.gov/ncbddd/hearingloss/ehdi-data2014.html
Clayton, E. W. (1992). Issues in state newborn screening programs. Pediatrics, 90, 641-646.
D'Agostino, J. A., & Austin, L. (2004). Auditory neuropathy: A potentially under-recognized neonatal intensive care unit sequela. Advanced Neonatal Care, 4, 344-353.
Fleischman, A. R., Post, L. F., & Dubler, N. N. (1994). Mandatory newborn screening for human immunodeficiency virus. Bulletin of the New York Academy of Medicine, 71, 4-17.
Harlor, Jr., A. D. B., & Bower, C. (2009). Hearing assessment in infants and children: Recommendations beyond neonatal screening. Pediatrics, 124, 1252-1263.
Health Insurance Portability and Accountability Act of 1996, Pub. L. No. 104-191, 110 Stat. 1938 (1996).
Health Resources and Services Administration. (2009). Enhancing communication: Improving care for infants with hearing loss. Washington, DC: U.S. Department of Health and Human Services.
Johnson, J., White, K., Widen, J., Gravel, J., James, M., Kennalley, T., . . . Holstrum, J. (2005). A multicenter evaluation of how many infants with permanent hearing loss pass a two-stage otoacoustic emissions/automated auditory brainstem response newborn hearing screening protocol. Pediatrics, 116, 663-672.
Joint Committee on Infant Hearing. (2007). Year 2007 position statement: Principles and guidelines for early hearing detection and intervention programs. Pediatrics, 120, 898-921.
Kirkim, G., Serbetcioglu, B., Erdag, T. K., & Ceryan, K. (2008). The frequency of auditory neuropathy detected by universal newborn hearing screening program. International Journal of Pediatric Otorhinolaryngology, 72, 1461-1469.
Mehl, A., & Esquivel, M. (2016). Medical home & EHDI: The importance of appropriate & timely screening, diagnosis, management, & follow up. In L. R. Schmeltz (Ed.), The NCHAM eBook: A resource guide for early hearing detection and intervention. Retrieved from http://infanthearing.org/ehdi-ebook/2016_ebook/7%20Chapter7MedicalHome2016.pdf [PDF]
National Center for Hearing Assessment and Management (NCHAM). (2008, May). The impact of privacy regulations: How EHDI, Part C, & health providers can ensure that children & families get needed services. Retrieved from https://www.infanthearing.org/coordinator_orientation/section13/45_privacy_regs.pdf [PDF]
New York State Department of Health, Early Intervention Program. (2007). Clinical practice guideline: Report of the recommendations. Hearing Loss, assessment and intervention for young children (age 0-3 years) [New York State Department of Health Pub. No. 4967]. Albany, NY: Author.
Russ, S. A., Hanna, D., DesGeorges, J., & Forsman, I. (2010). Improving follow-up to newborn hearing screening: A learning-collaborative experience. Pediatrics, 126, S59-S69.
Siegel, J. D., Rhinehart, E., Jackson, M., Chiarello, L., & Healthcare Infection Control Practices Advisory Committee. (2007). Guideline for isolation precautions: Preventing transmission of infectious agents in healthcare settings. Retrieved from https://www.cdc.gov/hicpac/pdf/isolation/Isolation2007.pdf
Turchetta, R., Orlando, M. P., Cammeresi, M. G., Altissimi, G., Celani, T., Mazzei, F., . . . Cianfrone, G. (2012). Modifications of auditory brainstem responses (ABR): Observations in full-term and pre-term newborns. The Journal of Maternal-Fetal & Neonatal Medicine, 8, 1342-1347. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/22122007
Vohr, B. R., Oh, W., Stewart, E. J., Bentkover, J. D., Gabbard, S., Lemons, J., . . . Pye, R. (2001). Comparison of costs and referral rates of 3 universal newborn hearing screening protocols. Journal of Pediatrics, 139, 238-244.
Yoshinaga-Itano, C., DeConde Johnson, C., Carpenter, K., & Stredler Brown, A. (2008). Outcomes of children with mild bilateral hearing loss and unilateral hearing loss. Seminars in Hearing, 29, 96-211.
Acknowledgements
Content for ASHA's Practice Portal is developed through a comprehensive process that includes multiple rounds of subject matter expert input and review. ASHA extends its gratitude to the following subject matter experts who were involved in the development of the Newborn Hearing Screening page:
- Kathryn L. Beauchaine, MA, CCC-A
- Brandt Culpepper, PhD, CCC-A
- Janet M. Farrell
- Jeffrey K. Hoffman, PhD, CCC-A
- Michelle A. King, AuD, CCC-A
- Patti F. Martin, PhD, CCC-A
- Ryan W. McCreery, PhD
- Mary Pat Moeller, PhD, CCC-A
- Beth A. Prieve, PhD, CCC-A
- Jackson Roush, PhD, CCC-A
- Diane L. Sabo, PhD, CCC-A
- Lynn G. Spivak, PhD, CCC-A
- Anne Marie Tharpe, PhD, CCC-A
In addition, ASHA thanks the members of the Newborn Hearing Screening Working Group whose work was foundational to the development of this content. Members of the working group included Beth Prieve (chair), Kathryn Beauchaine, Diane Sabo, Anne Marie Tharpe, and Anne Oyler (ex officio). ASHA Vice Presidents for Professional Practices in Audiology Jaynee Handelsman (2010-2012) and Donna Fisher Smiley (2013-2015) served as the monitoring officers.
ASHA also thanks the members of the Working Group on Loss to Follow-Up whose work was foundational to the development of this content. Members of the working group included Anne Marie Tharpe (chair), John Eichwald, Janet Farrell, Jeffrey Hoffman, Meredith Isola, Patti Martin, Amy M. Robbins, Lynn Spivak, Susan Wiley, and Pam Mason (ex officio). Gwendolyn D. Wilson, ASHA vice president for audiology practice (2007-2009), served as monitoring officer.
Citing Practice Portal Pages
The recommended citation for this Practice Portal page is:
American Speech-Language-Hearing Association (n.d). Newborn Hearing Screening. (Practice Portal). Retrieved month, day, year, from www.asha.org/Practice-Portal/Professional-Issues/Newborn-Hearing-Screening/.
Content Disclaimer: The Practice Portal, ASHA policy documents, and guidelines contain information for use in all settings; however, members must consider all applicable local, state and federal requirements when applying the information in their specific work setting.











